Assays for Vaccine Development and Testing
Best-in-Class Services for COVID-19, Flu, and Other Infectious Diseases
At Nexelis, a Q² Solutions Company, we approach each vaccine program with disciplined agility: scientific excellence and full transparency with the flexibility, ingenuity, and capacity for the quick decision-making needed. Our expertise is built on a foundation of 20 years of experience in vaccine efficacy and safety evaluation as a partner to industry leading vaccine manufacturers worldwide. Cutting-edge technology platforms and state-of-the-art laboratories enable us to best develop, qualify, and validate single and multiplex immunological assays and sample testing to support vaccine development.
Nexelis has seven integrated platforms that help you progress from vaccine development through clinical evaluation. Our main focus is on the evaluation of the efficacy of the vaccine — be it in the developmental stages or in clinical immunological responses. Critical reagents and well-established assays are indispensable to the efficient assessment of vaccine candidates. We take pride in our ability to develop innovative and critical reagents in-house. This capacity allows us to scale rapidly and helps control batch-to-batch variations of assay materials — crucial factors in maintaining operational excellence, high quality data, and world class science for our clients.

Our seven platforms are:
- Viral functional assays
- Bacterial functional assays
- Ligand binding assays
- Molecular biology
- Cellular assays
- Protein science
- In vivo models (preclinical)
Automation to execute our assays with each platform also further reduces operator error and increases throughput and assay precision.

Nexelis is the winner of Terrapinn’s 2022 Vaccine Industry Excellence (ViE) Award for Best Central/Specialty Laboratory.
Assays Supporting Vaccine Programs for Viral and Bacterial Diseases
Our experience covers assay development for a multitude of pathogens:
Respiratory
Seasonal and pandemic influenza, Haemophilus influenzae, respiratory syncytial virus (RSV), Neisseria meningitidis, Streptococcus pneumoniae, Bordetella pertussis, Corynebacterium diphtheriae, rhinovirus, SARS-CoV-2 and its variants

Enteric
Escherichia coli, Shigella spp., Clostridium difficile, rotavirus, Salmonella typhimurium

Vector/Zoonotic
Chikungunya, Zika, dengue, malaria, yellow fever

Sexually Transmitted
Cytomegalovirus, herpes, hepatitis B

Other Infectious Diseases
Human papilloma virus (HPV), adenovirus, adeno-associated virus (AAV), measles, mumps, rubella, varicella zoster, tetanus, Staphylococcus aureus, Streptococcus aga lactiae (GBS), Lymphocytic Choriomeningitis Virus (LCMV), mammarenavirus, vaccinia, etc.
Expertise Across All Types of Vaccines
Nexelis has years of experience in the development of a broad range of vaccine types, including:
- Live-attenuated vaccines
- Inactivated vaccines
- Subunit, recombinant, polysaccharide, and conjugate vaccines
- Toxoid vaccines
- Virus-like-particle (VLP) based vaccines
- DNA and RNA vaccines
- Adeno-Associated Virus (AAV) vector-based vaccines
- Lentivirus/Vesicular Stomatitis Virus (VSV)-based pseudo-particles
- Therapeutic vaccines
We believe in the power of partnership and strive to work as a seamless extension of your internal research lab.
Enzyme-Linked Immunosorbent Assay (ELISA)
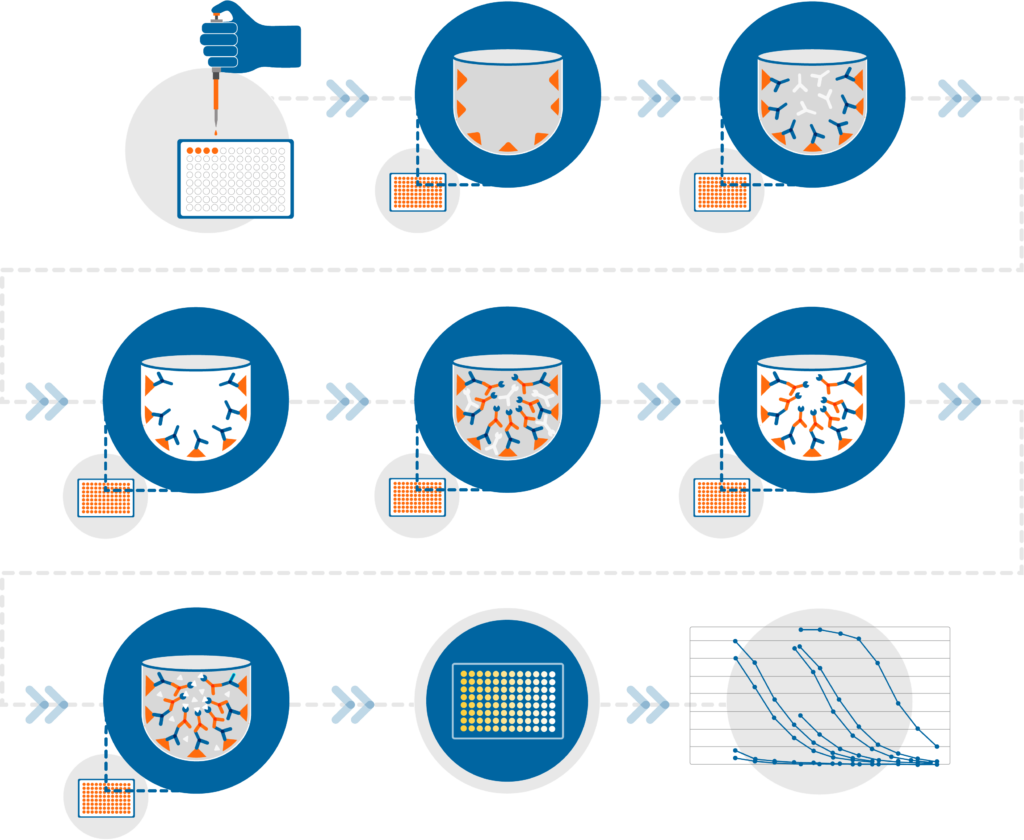
Scope of Vaccine Development Services
At every stage of your vaccine development project — whether it is lead finding, target validation, or assessment of efficacy or immunogenicity — a reliable assay is the key to success. Choosing the right set of assays is critical to obtain meaningful data. Several well-designed and optimized assays are required at every stage to generate reliable and predictive data to translate vaccine development programs from early discovery through preclinical, clinical, and regulatory approval.
Using an agile approach, Nexelis develops and validates a broad range of assays that are robust and reproducible. Employing these methods using our high-throughput automated instruments increases our testing capacity, reduces cost, improves operational efficiency, and delivery times — all custom-tailored to the ideal specifications of your project. This fit-for-purpose approach, acting as an extension of the lab with our platform solutions, allows us to provide a comprehensive, individualized solution. Choose Nexelis for your vaccine development and baseline vaccine evaluation requirements.
Nexelis continuously invests in high-quality research and development to further enable our portfolio. Our recent acquisition of GSK Vaccines’ Marburg, Germany, clinical bioanalytical laboratory along with a laboratory in China strengthens our ability to rapidly develop effective vaccines for several life-threatening infections.
